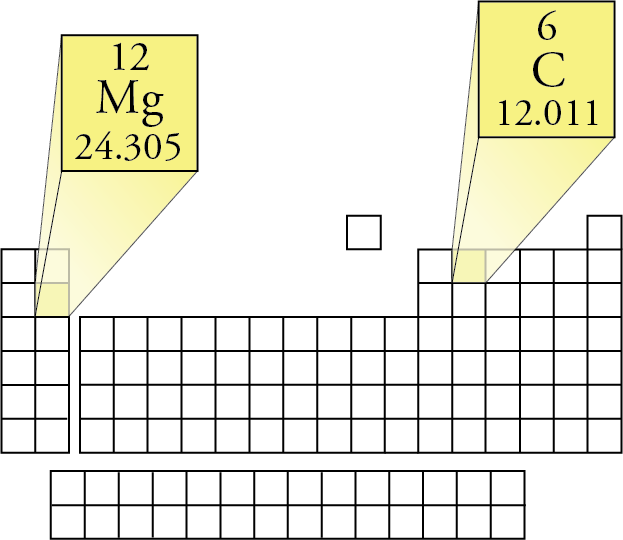
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994.
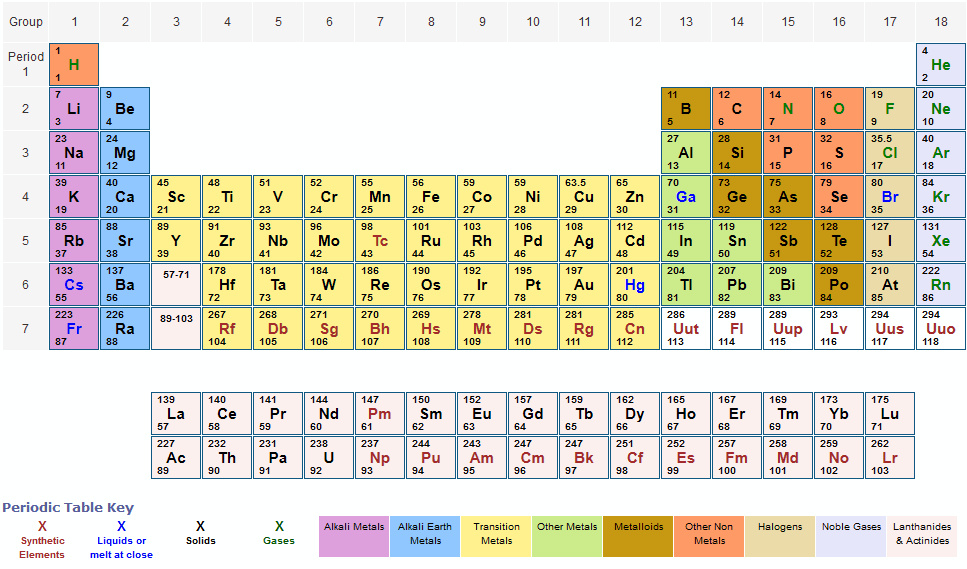
Find atomic masses: look up the atomic masses of each element present in the compound. First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules. The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents.